Solid-Liquid Interface-Driven Functionality
The structure of electrical double layers (EDL) at electrified interfaces is of utmost importance for electrochemical energy storage as well as printable, flexible, and bio-electronic devices, such as ion-gated transistors. Ionic liquids (IL) have attracted considerable attention not only due to their large electrochemical window but also due to the high energy charge density they can provide.
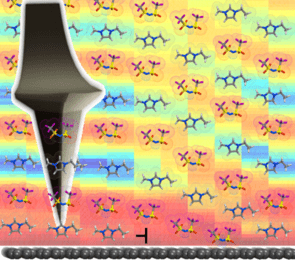
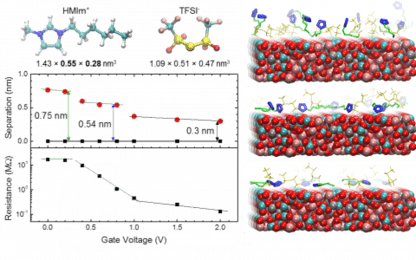
This is, for example, used to modify the current-voltage characteristics of metal oxides due to electrostatic doping or modification of oxygen vacancy concentrations. While this is explored on the device level, connections to the molecular level structure of the EDL are largely missing. An important consideration is the existence of defects in the ion layering observed in the EDL of IL and its consequences for charge storage or gating properties. Due to the fact that an AFM probe is sensitive to the mass density changes when approaching the solid interface inside the liquid, this approach can be used to image the EDL structure ad map it in three dimensions under in situ operation of electrochemical capacitors or gated transistors.
This approach shines a light on the correlation of molecular length scale processes and the macroscopically measured device performance. This concept can be expanded to use solid-liquid interfaces to modify polar materials and their functionality. The use of a liquid provides a well-defined chemical potential at the solid-liquid interface, which can provide additional screening mechanisms for polar materials, such as ferroelectrics. The use of a liquid has the advantage of electrochemical control of surface chemistry and thus order parameter tuning.
Since AFM can be used to study piezoelectric and ferroelectric properties in liquid, this platform offers a unique opportunity to link the interfacial structure of the liquid and underlying polar properties.
Highlights